|
Abstract
Background:
Older patients are now the rule rather than the
exception in hemodialysis (HD). Cognitive impairment
is common among persons with end stage renal disease
(ESRD) and is associated with poor outcomes.
Aim:
To assess the prevalence of cognitive impairment
among older adults on HD and the association of
different patients' demographics and characteristics
with cognitive impairment.
Materials and methods:
A cross section study was conducted to assess
the cognitive functions of elderly subjects on
HD. 94 subjects, 60 years old and above, were
included. All subjects were subjected to: 1- comprehensive
geriatric assessment. 2- Laboratory investigations
including: hemoglobin, serum creatinine, serum
urea, serum potassium, serum sodium and serum
albumin. Kt/V as a marker of dialysis adequacy
was calculated.
Results: The study
revealed that 26 (27.7%) patients had normal cognitive
function, 32 (43%) had mild cognitive impairment,
21 (23%) had mild dementia, 8 (8.5%) had moderate
dementia and 7 (7.4%) had severe dementia. Older
age, low education level and longer duration of
dialysis history were found to have significant
associations with cognitive impairment, [P: <
0.001, 0.002, and 0.012 respectively]. While hemoglobin,
serum albumin, serum creatinine, serum urea, serum
potassium and Kt/V showed no significant association
with cognitive impairment, Serum sodium and dry
weight dialysis showed significant difference
between patients with normal cognitive function
and patients with cognitive impairment, [P <0.001
both].
Conclusion: Cognitive
impairment is prevalent in Egyptian older adults
on HD and more prevalent and severe in those with
older age, low education, lower level of serum
sodium, longer duration of dialysis history and
higher dry weight.
Key words: Cognitive
impairment, Egypt, Hemodialysis
|
- - - - - - - - - - - - - - - - - - -
- - - - - - - - - - - - - - - - - - - - - - - -
Introduction
Older patients are now the rule rather than the exception
on hemodialysis (HD). According to the United States
Renal Data System (USRDS) data from 2006, nearly one
half of incident dialysis patients in the United States
are senior citizens, with the median age at dialysis
initiation at 64.4 years
old. Furthermore, the elderly are the fastest-growing
group of incident dialysis patients, meaning that this
median age will continue to increase. Nearly all of
these elderly patients employ HD, with only 3 to 5%
using peritoneal dialysis (PD). (1)
Cognitive impairment is common
among persons with end stage renal disease (ESRD) and
is associated with poor outcomes, (2) but its underlying
mechanisms remain poorly understood. As a result, few
evidence-based strategies exist for treating this serious
morbidity. Uncontrolled studies have reported improvements
in cognitive function after kidney transplantation and
more recently, after conversion from conventional to
nocturnal hemodialysis, suggesting that modifiable factors
associated with ESRD and/or its treatment may be implicated
in the pathogenesis of this disorder. (3)
Many studies found that cognitive impairment was prevalent
in older adults with CKD on HD as one reported that
37 percent of patients had severe cognitive impairment.
(4) And another study found that 38 percent had severe
impairment in executive function and 33 percent severe
memory impairment. (5)
Several ESRD- and dialysis-associated
factors such as retention of uremic solutes, hypertension,
hemodynamic instability during dialysis, and anemia
may be favorably modified by more frequent hemodialysis
schedules. Several of these conditions have also been
implicated in the pathogenesis of cognitive impairment.
For example, in the National Cooperative Dialysis Study,
higher levels of urea clearance were correlated with
better cognitive performance. (6) In addition, cardiovascular
and hemodynamic factors (hypertension, and at the other
extreme, intra dialysis hypotension) may lead to stroke
or cerebral ischemia and contribute to cognitive impairment
in patients with ESRD. (7)
The etiology of cognitive impairment
among HD patients is thought to be multifactorial, and
includes factors such as cerebrovascular lesion, (7)
hypotension, (8) abnormalities of serological data,
(9) social history, (10) and e GFR level; (11) as, each
10 mL/min/1.73 m2 decrement in e GFR was found to correspond
to an approximately 15 to 25 percent increase in the
risk of cognitive dysfunction among individual cognitive
domains. (12) In addition, the high prevalence of cardiovascular
risk factors might overshadow the roles of aging and
non-vascular factors in the development of cognitive
impairment. (13)
Dementia is associated
with an increased risk of multiple adverse outcomes.
Prevalent dementia in hemodialysis patients increases
the risk of hospitalization. Dementia also increases
costs of care; in 2002 approximately $19,100 more Medicare
dollars were spent over one year in hemodialysis patients
with dementia compared to those without. (14) Among
hemodialysis patients, dementia was associated with
a 1.48 fold increased risk of death over one year. (10)
In this study we assessed the prevalence of cognitive
impairment among older adults on HD and association
between cognitive impairment and different demographics
and characteristics of these patients.
Subjects
and Methods
Study setting and sample:
• A cross section study was conducted to assess
the cognitive functions in elderly subjects on HD. 94
elderly subjects 60 years old and above (Both males:
n=39, and females: n= 55) were recruited from Ain Shams
University hospitals.
Exclusion criteria:
• The study did not include subjects with stroke,
delirium, alcoholism or drug abuse, psychiatric disease,
thyroid disease and auditory or visual impairment were
excluded as these conditions are known to affect cognitive
functions. Also subjects with chronic liver disease
and Chronic Obstructive Pulmonary Disease (COPD) were
excluded.
Data collection:
All subjects were subjected to:
A. Informed oral consent.
B. Comprehensive geriatric assessment, including
1. Medical history and physical examination.
2. The Mini-Mental Status Examination MMSE was applied
to all the participants to assess their cognitive function.
(15)
All subjects were screened for presence of dementia
by using the Arabic version of MMSE. (16)
The MMSE was selected because it is the best studied
instrument for screening for dementia. (17)
The MMSE comprises 30 questions with 10 devoted to orientation
(five regarding time and five regarding place). Three
items requiring registration of new information (repeating
three words). Five questions addressed attention and
calculation. Mental control questions requiring patient
to make five serial subtractions of 7 from 100 or spell
word backward); three recall items (remembering the
three registration items), eight items assessing language
skills (two naming items, repeating phrase, following
a three-step command, reading and following a written
command and writing a sentence), and one construction
question (copying a figure consisting of two overlapping
pentagons) were used.
According to the MMSE, the subjects were classified
into two groups:
Group 1 (controls): cognitively intact: MMSE
= 30
Group 2 (cases): cognitively impaired: MMSE <
30
And according to the severity of cognitive impairment,
participants in group 2 were further classified into
4 subgroups:
Group a: mild cognitive impairment: MMSE: 26 - 29.
Group b: early dementia: MMSE: 21 - 25.
Group c: moderate dementia: MMSE: 11 - 20.
Group d: severe dementia: MMSE: 0 - 10.
C. Laboratory Investigations
including:
• Hemoglobin
(g\dl)
• Serum creatinine
(mg\dl)
• Serum urea
(mg\dl)
• Serum potassium
(mmol\L)
• Serum sodium
(mmol\L)
• Serum albumin
(g\dl)
• Serum blood
urea nitrogen (mg/dl) pre-dialysis and post-dialysis
D. Dialysis adequacy:
Kt/V as a marker of dialysis adequacy
Kt/V is a number used to quantify hemodialysis treatment
adequacy.
• K - dialyzer
clearance of urea
• t - dialysis
time
• V - volume
of distribution of urea, approximately equal to patient's
total body water
In the context of hemodialysis, Kt/V is a pseudo-dimensionless
number; it is dependent on the pre- and post-dialysis
concentration. It is not the product of K and t divided
by V, as would be the case in a true dimensionless number.
(18)
It was developed by Frank Gotch and John Sargent as
a way of measuring the dose of dialysis when they analyzed
the data from the National Cooperative Dialysis Study.
(19) In hemodialysis the US National Kidney Foundation
Kt/V target is ? 1.3, so that one can be sure that the
delivered dose is at least 1.2 (20)
Calculation of Kt/V needs serum blood urea nitrogen
(mg/dl) pre-dialysis and post-dialysis, weight of the
patient pre-dialysis and post-dialysis, treatment time
(minute) and frequency of treatments/week. With these
parameters Kt/V is calculated by using an online calculator:
http://www.davita.com/ktvcalculator/
Statistical Methods:
All data were entered into the 21st version of SPSS
(Statistical Package of Social Science) and analyzed
using frequency and descriptive statistics to analyze
the study population. Frequency and percentage was calculated
for all qualitative variables. Description of all data
in the form of mean (M) and standard deviation (SD)
was done for all quantitative variables. Comparison
of qualitative variables was done using Chi-square test;
it is a test that determines the extent that a single
observed series of proportions differs from a theoretical
or expected distribution of proportions, or the extent
that two or more series of proportions or frequencies
differ from one another based on the chi-square distribution.
Comparison between quantitative variables was done using
ANOVA (analysis of variance) to compare between more
than two groups.
The level of significance for Chi-square test and ANOVA
was taken at P value < 0.05 is significant, otherwise
it is non-significant.
Results
Table 1: Patient's demographics and characteristics:
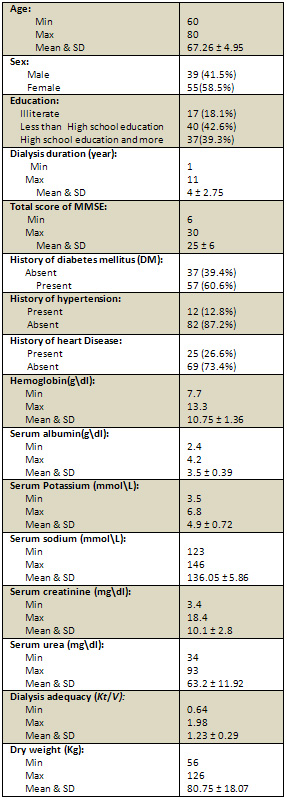
The study was conducted on 94 older patients on regular
haemodialysis. Table 1 shows the demographics and characteristics
of the older adults: The mean age of the sample was
67.26 ± 4.95 (range: 60 - 80). The sample included
39 (41.5%) males and 55 (58.5%) females. Among the 94
older subjects; 17 (18.1%) were illiterate, 40 (42.6%)
had less than high school education and 37 (39.3%) had
high school education or more. The mean dialysis duration
in years was 4 ± 2.75 (range: 1 - 11). The mean
of Total score of MMSE was 25 ± 6 (rang 30-6).
37 (39.4%) subjects had no history of DM and 57 (60.6%)
had history of DM. 25 (26.6%) subjects had history of
heart disease and 69 (73.4%) did not. 12(12.8%) subjects
had history of HTN and 82 (87.2%) had no HTN. The mean
hemoglobin level was 10.75 ± 1.36 (range 7.7
- 13.3). The mean level of serum albumin was 3.5 ±
0.39 (range 2.4 - 4.2). The mean level of serum potassium
was 4.9 ± 0.72 (range 3.5 - 6.8). The mean level
of serum sodium was 136.05 ± 5.86 (range 123
- 146). The mean level of serum creatinine was 10.1
± 2.8 (range 3.4 - 18.4). The mean level of serum
urea was 63.2 ± 11.92 (range 34 - 93). The mean
value of dialysis adequacy (Kt/V) was 1.23 ±
0.29 (range 0.64 - 1.98). The mean value of dry body
weight (Kg) 80.75 ± 18.07 (range 56 - 126).
Figure 1:
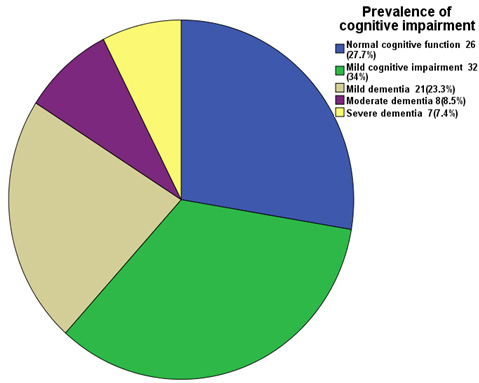
Figure 1 shows that 26
(27.7%) patients had normal cognitive function, 32 (43%)
had mild cognitive impairment, 21 (23%) had mild dementia,
8 (8.5%) had moderate dementia and 7 (7.4%) had severe
dementia.
Click here for Table
2: Relationship between Patients' characteristics and
cognitive impairment:
Table 2 shows the association of patients' demographics
and characteristics with cognitive function; the mean
age of the patients with normal cognitive function was
67.1 ± 3.16, while for patients with severe dementia
was 73.85 ± 3.33, [P:<0.001]. Sex did not
show a significant difference between patients with
normal cognitive function and patients with cognitive
impairment [P: 0.35]. Also history of DM, HTN heart
disease did not show significant association with cognitive
function [P: 0.21, P: 0.87, P: 0.82 respectively]. The
education level and the duration of dialysis history
showed significant differences between patients with
normal cognitive function and patients with cognitive
impairment, [P: 0.002, P: 0.012 respectively]. While
hemoglobin level, serum albumin, serum creatinine, serum
urea and serum potassium showed no significant difference
between patients with normal cognitive function and
patients with cognitive impairment, [P: 0.099, P: 0.17,
P: 0.18, P: 0.08, P: 0.35 respectively]. Serum sodium
showed significant difference between patients with
normal cognitive function and patients with cognitive
impairment, [P:< 0.001]. And also Dry weight dialysis
showed significant difference between patients with
normal cognitive function and patients with cognitive
impairment, [P :< 0.001], while dialysis adequacy
(Kt/v) showed no significant difference between patients
with normal cognitive function and patients with cognitive
impairment, [P: 0.79].
Discussion
The purpose of this study was to determine prevalence
of cognitive impairment among older adults on HD and
to assess the association between the prevalence and
severity of cognitive impairment with different demographics
and characteristics of these patients.
Despite the growing numbers of patients with ESRD and
dementia, the medical literature did not truly explore
the intersection of these two groups of patients. Recently,
Seliger et al, 2004 [21] reported that elevated serum
creatinine was associated with a higher risk for dementia
in older adults who reported either good or excellent
health.
In our study we found that prevalence
of cognitive impairment among older adults on HD was
72.3 % (43% had mild cognitive impairment, 23% had mild
dementia, 8.5% had moderate dementia and 7.4% had severe
dementia).
This result was supported by Kurella et al, 2004 [5]
who studied 80 HD patients (mean age 61.2 ± 14.3
years) and found that prevalence of severe memory impairment
was 33 %.
Also Tyrrell et al, 2005 [22]
found that in older adult patients on HD the prevalence
of cognitive impairment, based on the Mini-Mental State
Examination (MMSE), was 47% of 51 HD outpatients.
This was also supported by Gen
et al, 2011 [23] who found that the prevalence of cognitive
impairment based on the MMSE was 18.8% in HD patients.
They stated that HD patients had showed a higher prevalence
of cognitive impairment in older groups (50 years and
older).
Also we agreed with another
study conducted by Murray et al, 2006 [4] to assess
the cognitive function across multiple cognitive domains
in 338 HD patients (mean age, 71.2 ± 9.5 years)
and found that among older adults on HD, 13.9% were
classified with mild impairment, 36.1% with moderate
impairment, 37.3% with severe impairment, and 12.7%
with normal cognition.
The prevalence of severe cognitive impairment in some
studies was higher than our study as the authors included
patients with history of cerebrovascular diseases. But
in our study we excluded them and other diseases that
could directly affect cognitive function e.g. alcoholism,
drug abuse, psychiatric disease, thyroid disease, auditory
or visual impairment, chronic liver disease and chronic
obstructive pulmonary disease (COPD).
Rakowski et al, 2006 [24] and
Laudanski et al, 2010 [25] reported that in patients
undergoing HD, cognitive impairment brought more serious
consequences, such as hospitalization and reduced life
expectancy. They also added that cognitive impairment
in HD patients might hinder them from complying with
dialysis schedules, medications, and dietary restrictions
Also our study revealed that
cognitive impairment was more prevalent and more severe
in patients with older age, lower education level, longer
duration of hemodialysis, lower level of serum sodium
and higher dry weight.
This was supported by Gen et
al, 2011 [23] who found that among HD patients, level
of education was associated with MMSE score, and added
that serum sodium level, dry weight and history of cerebrovascular
disease tended to be associated with low MMSE score
of HD patients.
Also Murray et al, 2006 [4] who assessed the cognitive
function across multiple cognitive domains in 338 HD
patients, found that low education was associated with
severe cognitive impairment.
Sehgal et al, 1997 [26] stated
that older age and lower education level were independently
associated with less than 24 points achievement on MMSE
test in multivariable analysis.
As regards hyponatremia, Gen
Odagiri1 et al, 2011 [27] found a significant relationship
[P = 0.05] between hyponatremia and the MMSE score among
HD patients; they added that this relationship was also
reported in a previous study by Maugeri et al. 1999
[28]
Hyponatremia depends on various factors including blood
dilution by chronic fluid overload and dietary sodium
restriction. [29] These factors might explain why no
correlations were observed with other blood parameters,
which were, to some extent, stabilized by HD.
The relationship between higher dry weight and cognitive
impairment could be explained by the known relationship
between obesity and cognitive impairment as reported
by Gustafson et al, 2003 [30] who found an association
between greater BMI at age 70 and greater risk of incident
dementia in an 18-year longitudinal observational study.
Our result also showed although the mean levels of serum
urea and serum creatinine were higher in patients with
severe cognitive impairment than patients with normal
cognitive function, this was statistically insignificant.
This can be explained by the relatively small sample
size.
Also the results showed that although dialysis adequacy
(mean of Kt/v) was high in patients with normal cognitive
function than those with cognitive impairment this was
statistically insignificant. This can be explained as
the dialysis adequacy (mean of Kt/v) for the whole sample
(with and without cognitive impairment) was satisfactory.
This study was supported by
Dahbour et al, 2009 [31] who studied and compared the
predialysis (PrHDSc) and postdialysis (PoHDSc) mini
mental status examination score Patients' (PrHDSc) correlated
positively with PoHDSc and dialysis efficiency measured
by Kt/V (r=0.58, 0.4, and 0.34, respectively). Education
level correlated positively with PrHDSc r=0.41 but not
PoHDSc.
Conclusion
Cognitive impairment was prevalent in Egyptian older
adults on HD and more prevalent and severe in those
with older age, lower education, longer history of hemodialysis
duration, lower level of serum sodium and higher dry
weight. So we recommended including cognitive evaluation
in assessment of older adults on HD and conduction of
of a larger sample size case control study to identify
modifiable risk factors of cognitive impairment in older
adults in HD.
References
1. U.S. Renal Data System: USRDS 2008 Annual Data Report:
Atlas of Chronic Kidney Disease and End-Stage Renal
Disease in the United States. Bethesda, MD, National
Institute of Diabetes and Digestive and Kidney Diseases,
2008.
2. Kurella M, Mapes DL, Port FK, Chertow GM.:
Correlates and outcomes of dementia among dialysis patients:
The Dialysis Outcomes and Practice Patterns Study. Nephrol
Dial Transplant, 2006; 21: 2543 - 2548.
3. Griva K, Thompson D, Jayasena D, Davenport A, Harrison
M, Newman SP.: Cognitive functioning pre- to post-kidney
transplantation-A prospective study. Nephrol Dial Transplant,
2006; 21: 3275-3282.
4. Murray AM, Tupper DE, Knopman DS, et al. Cognitive
impairment in hemodialysis patients is common. Neurology,
2006; 67:216-223.
5. Kurella M, Chertow GM, Luan J, et al. Cognitive impairment
in chronic kidney disease. J Am Geriatr Soc, 2004; 52:1863-1869.
6. Teschan PE, Bourne JR, Reed RB, Ward JW. Electrophysiological
and neurobehavioral responses to therapy: The National
Cooperative Dialysis Study. Kidney Int Suppl, 1983;
13: S58-S65.
7. Prohovnik I, Post J, Uribarri J, Lee H, Sandu O,
Langhoff E.: Cerebrovascular effects of hemodialysis
in chronic kidney disease. J Cereb Blood Flow Metab,
2007; 27: 1861-1869.
8. Mizumasa T, Hirakata H, Yoshimitu T, Hirakata E,
Kubo M, Kashiwagi M, Tanaka H, et al: Dialysis-related
hypotension as a cause of progressive frontal lobe atrophy
in chronic hemodialysis patients: a 3-year prospective
study. Nephron Clin Pract, 2004; 97:c23-c30.
9. Rakowski DA, Caillard S, Agodoa LY, Abbott KC: Dementia
as a predictor of mortality in dialysis patients. Clin
J Am Soc Nephrol, 2006; 1:1000-1005.
10. Kurella M, Mapes DL, Port FK, Chertow SF: Correlates
and outcomes of dementia among dialysis patients: the
dialysis outcomes and practice patterns study. Nephrol
Dial Transplant, 2006; 21:2543-2548.
11. Hailpern SM, Melamed ML, Cohen HW, et al. Moderate
chronic kidney disease and cognitive function in adults
20 to 59 years of age: Third National Health and Nutrition
Examination Survey (NHANES III). J Am Soc Nephrol, 2007;18:2205-2213.
12. Kurella M, Yaffe K, Shlipak MG, et al. Chronic kidney
disease and cognitive impairment in menopausal women.
Am J Kidney Dis, 2005; 45:66-76.
13. Arvanitakis Z, Wilson RS, Bienias JL, Evans DA,
Bennett DA: Diabetes mellitus and risk of Alzheimer
disease and decline in cognitive function. Arch Neurol,
2004; 61:661-666.
14. United States Renal Data System: USRDS. Bethesda,
MD: National Institutes of Health, National Institute
of Diabetes and Digestive and Kidney Diseases; 2005.
2005 Annual Data Report: Atlas of End-Stage Renal Disease
in the United States.
15. Folstein MF, Folstein SE , Mc Hug PR. Minimental
state. A practical method for grading the cognitive
state of patients for clinicians. J of Psychiat. Res.
1975; 12(3):189-98.
16. El-Okl MA, El Banouby MH , El Etrebi A. Prevalence
of Alzheimer dementia and other causes of dementia in
Egyptian elderly. MD Thesis, 2002; Faculty of Medicine,
Ain Shams University.
17. United States Preventive Services Task Force. Screening
for Dementia: Recommendation and Rationale. Ann Intern
Med. 2003; 138(11):925-926.
18. Bonert, M.; Saville, BA. "A non-dimensional
analysis of hemodialysis." Open Biomed Eng J, 2010;
4: 138-55.
19. Gotch FA, Sargent JA. "A mechanistic analysis
of the National Cooperative Dialysis Study (NCDS)".
Kidney int., 1985; 28 (3): 526-34.
20. "Clinical practice guidelines for nutrition
in chronic renal failure. K/DOQI, National Kidney Foundation."
Am J Kidney Dis, 2000; 35: S1-140.
21. Seliger SL, Siscovick DS, Stheman-Breen CO, Gillen
DL, Fitzpatrick A, Bleyer A, Kuller LH: Moderate renal
impairment and risk of dementia among older adults:
The Cardiovascular Health Cognition Study. J Am Soc
Nephrol, 2004; 15: 1904-1911.
22. Tyrrell J, Paturel L, cadec B, Capezzali E, Poussin
G: Older patients undergoing dialysis treatment: cognitive
functioning, depressive mood and health-related quality
of life. Aging Ment Health, 2005; 9:374 - 379.
23. Gen Odagiri1, Norio Sugawara1, Atsuhiro Kikuchi1,
Ippei Takahashi, Takashi Umeda, Hisao Saitoh, Norio
Yasui-Furukori1 et al: Cognitive function among hemodialysis
patients in Japan. Annals of General Psychiatry, 2011;
10: 20.
24. Rakowski DA, Caillard S, Agodoa LY, Abbott KC: Dementia
as a predictor of mortality in dialysis patients. Clin
J Am Soc Nephrol, 2006; 1:1000-1005.
25. Laudanski K, Nowak Z, Wa?kowicz Z: Psychological
aspect of dialysis: does cognitive appraisal determine
the overall outcome? Pol Arch Med Wewn, 2010; 120:49-52.
26. Sehgal AR, Grey SF, DeOreo PB, Whitehouse PJ. Prevalence,
recognition, and implications of mental impairment among
hemodialysis patients. Am J Kidney Dis, 1997; 30: 41-49.
27. Gen Odagiri, Norio Sugawara, Atsuhiro Kikuchi, Ippei
Takahashi, Takashi Umeda, Hisao Saitoh, Norio Yasui-Furukori
et al: Cognitive function among hemodialysis patients
in Japan. Annals of General Psychiatry, 2011; 10: 20.
28. Maugeri D, Malaguarnera M, Panebianco P, Balbagallo
P, Curasi MP, Bonanno MR, Spesiale S, et al: Assessment
of cognitive and affective disorders in an elderly population
undergoing hemodialysis. Arch Gerontol Geriatr, 1999;
29: 239-248.
29. Penne EL, Levin NW, Kotanko P: Improving volume
status by comprehensive dietary and dialytic sodium
management in chronic hemodialysis patients. Blood Purif,
2010; 30:71-78.
30. Gustafson D, Rothenberg E, Blennow K et al. An 18-year
follow-up of body mass index and risk for Alzheimer's
disease. Arch Intern Med, 2003; 163:1524-1528.
31. Dahbour SS, Wahbeh
AM, Hamdan MZ: Mini mental status examination (MMSE)
in stable chronic renal failure patients on hemodialysis:
the effects of hemodialysis on the MMSE score. A prospective
study. Hemodial Int, 2009; 13:80-85.
|

